This page was generated from `/home/lectures/exp3/source/notebooks/L27_AMA/L27_harmonic_oscillator.ipynb`_.
The harmonic oscillator¶
One of the most important examples in all branches of physics is the harmonic oscillator. The potential has the shape of a parabola with the potential energy of
and the repelling force depends linearly on the deviation from the equilibrium position
Fig.: A particle within the parabolic potential of a harmonic oscillator.
In the case of classical mechanics we allready know examples like a weight at a pendulum or a weight at a spring. We can treat the weight as a point-like mass
In order to discuss a quantum mechanic harmonic oscillator we start with the Schrödinger equation and the harmonic potential
which becomes
with the aid of the relation between the freuency
Now we introduce the substitutions
and reformulate the Schrödinger equation like
In the case of very high values of
If we now use the general solution within the re-formed Schrödinger equation, we get
The very last equation is a second order ordinary differenctial equation, which has the shape of a Hermite differential equation. Its solutions are the Hermite polynomials of degree
with
The first four Hermite polynomials
0 |
|||
1 |
|||
2 |
|||
3 |
In addition, the normalization factors
We can reformulate the Hermite polynomials through a power series expansion
This series has to be finite, since otherwise
cannot be normalized for all
Because the series of
wich results in
If we re-substitute
with
Since the quantum number
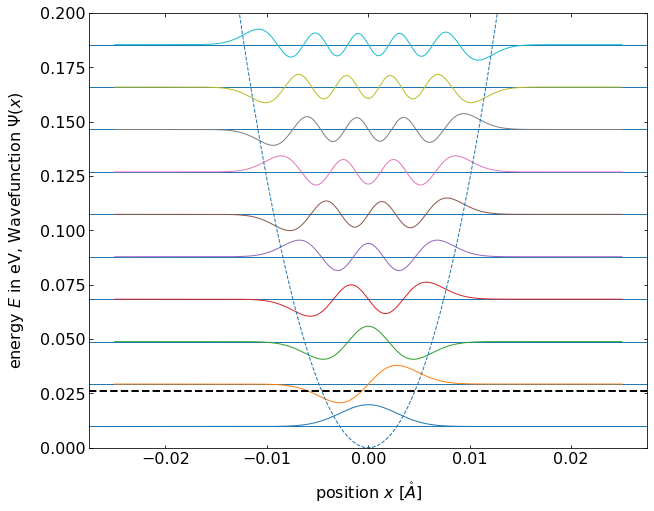
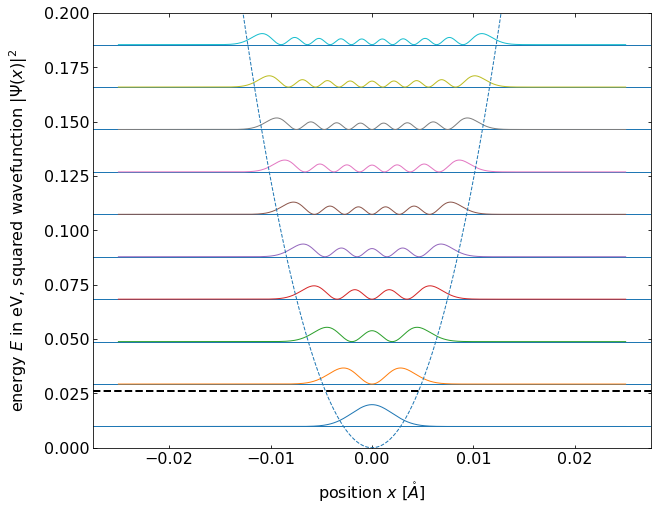
Fig.: (left) Wave functions :math:`psi_{mathrm{n}}` positioned at the height corresponding to the equidistant energy eigenvalue :math:`E_{mathrm{n}}`. (right) The according probability densities (squared wave functions). The postential is :math:`V = 0.5 cdot x^2`.
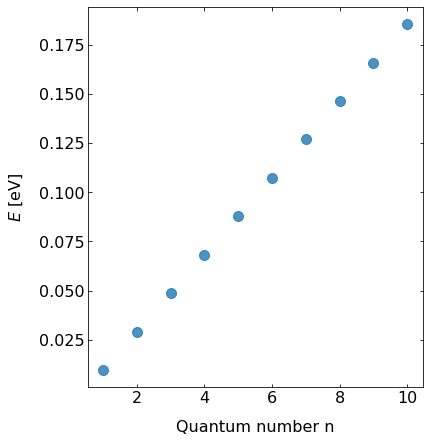
Fig.: The energy eigenvalues :math:`E_{mathrm{n}}` in dependence of the oscillation quantum number :math:`n` on a linear scale. The postential is :math:`V = 0.5 cdot x^2`.
Concerning classical mechanics, the probability to find the “classical” oscillating particle within the interval
where
At the state of lowets energy
If we consider higher quantum numbers, the probability density
Please note the squared wavefunction represents the probability density of stationary states. If one is seeking for dynamics, one has to consinder wave packets. The time dependence is related to the factor
